Research
Our research focuses on how modern therapeutic materials interact with the innate immune system and how these responses can be predicted, controlled, and exploited to improve human health. As advanced therapeutics become increasingly complex, ranging from nanoparticle drug delivery systems to nucleic acid platforms and biologically derived materials, understanding their immunological interface is essential for safe and effective translation.
A central theme of our work is programmable innate immunity. We investigate how innate immune responses can be shaped by therapeutic design, microbial signals, and cellular context. Through studies including, but not limited to, pattern recognition receptor signalling, innate immune pathways, inflammasome activation, complement pathways, bioenergetics, and epigenetic regulation, we aim to understand how innate immune cells become functionally reprogrammed and how these processes influence responses to infection, non-communicable disease, vaccines, gene therapies and advanced medicines to provide sustained innate immune protection and function.
Our work also explores the role of immunometabolism, examining how metabolic and bioenergetic states influence innate immune function and contribute to interindividual variability in immune responses. By linking immune signalling pathways with cellular metabolism, we seek to understand how immune responses emerge from the integrated behaviour of complex biological systems.
Using advanced immunological, cellular, and analytical approaches, we study a wide range of therapeutic platforms, including lipid nanoparticles, polymeric materials, virosomes, and exosomes, used for the delivery of diagnostics, small molecules, biologics, and nucleic acids.
Together, this work aims to define the principles governing immune responses to advanced therapeutics and to support the rational design of safer, more effective medicines.
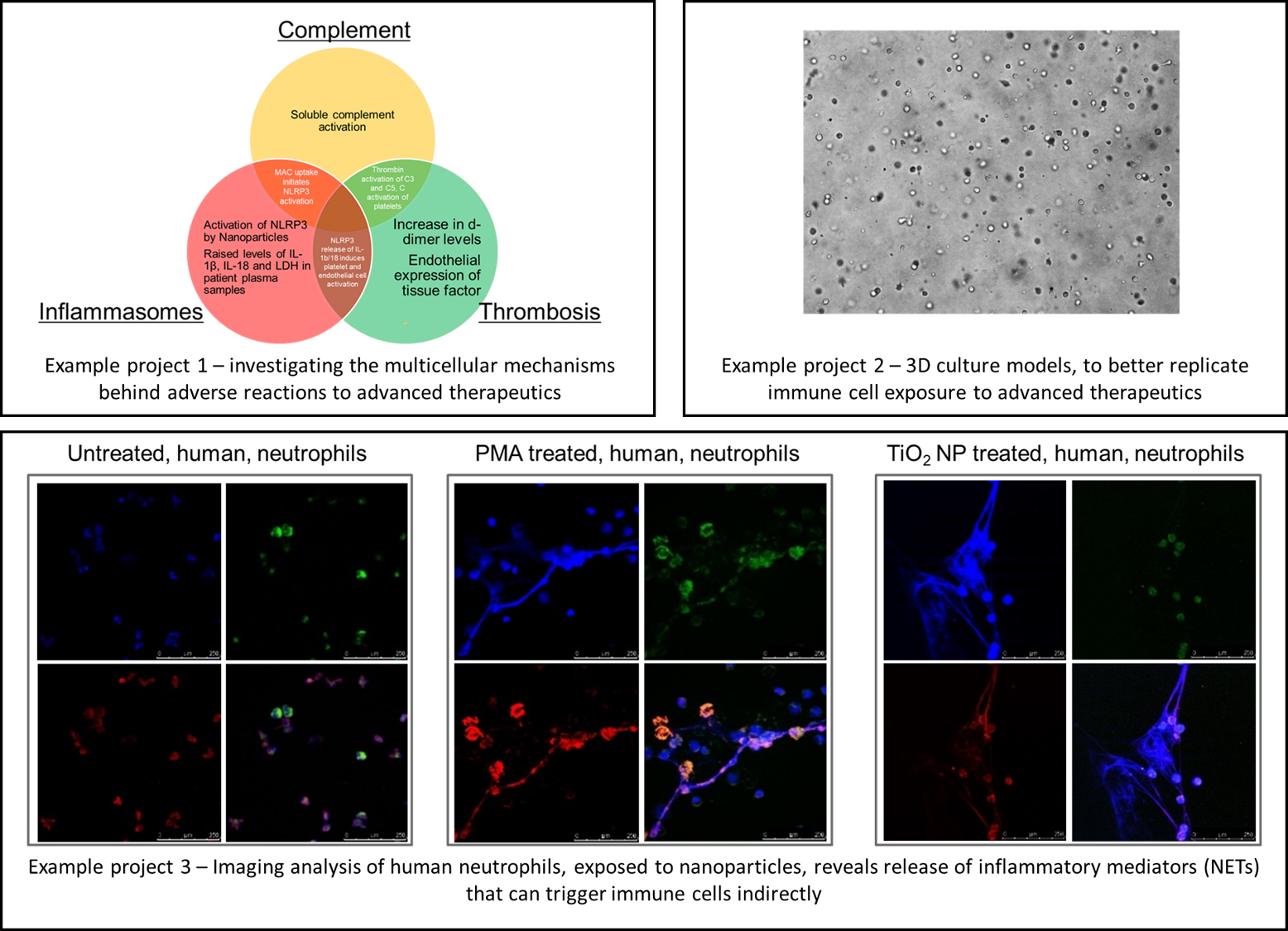
Immunocompatibility of Complex Medicines, Advanced Therapeutics, and Biomaterials
Our core question: How do modern drug delivery systems interact with innate immune pathways and what determines their safety?
Modern therapeutics increasingly rely on complex delivery systems, including nanoparticles, lipid carriers, viral vectors, and long-acting drug depots. While these technologies enable new treatment strategies, they also interact directly with the innate immune system.
Our research investigates how advanced therapeutics and biomaterials are recognised by immune cells and how these interactions influence safety, efficacy, and clinical translation. We study key innate immune pathways involved in these responses, including complement activation, inflammasome signalling, and cytokine release.
This work aims to define the principles governing the biocompatibility of complex medicines, enabling the rational design of therapeutic systems that elicit the desired immune response while avoiding unwanted inflammation or toxicity.
Key areas include:
Nanomedicine and lipid nanoparticle delivery systems
Long-acting therapeutics and drug depots
Biomaterial–immune interactions
Complement activation and inflammasome signalling
Immunocompatibility and immune-mediated toxicity
Keywords: advanced therapies and therapeutics, liposomes, lipidic nanoparticles (LNP), CARPA, pseudoallergy, hypersensitivity, complex medicines, extracellular vesicles, nanomedicines, nanotherapeutics, biocompatibility, immunology, immunotoxicology, intracellular drug delivery (ICD)
Sub-project;
- Development of techniques and technologies for the characterisation of nanomaterials.
The field of nanoimmunotoxicology is constantly evolving as new materials are developed. To meet these challenges, it is necessary to ensure that current techniques and methodologies are sufficient to identify potentially undesirable nanomaterial interactions with biological systems. New characterisation techniques are studied for their utility and introduced into our assay cascade as appropriate. Interaction with agencies such as EMA and FDA is also key to staying ahead of regulatory requirements.
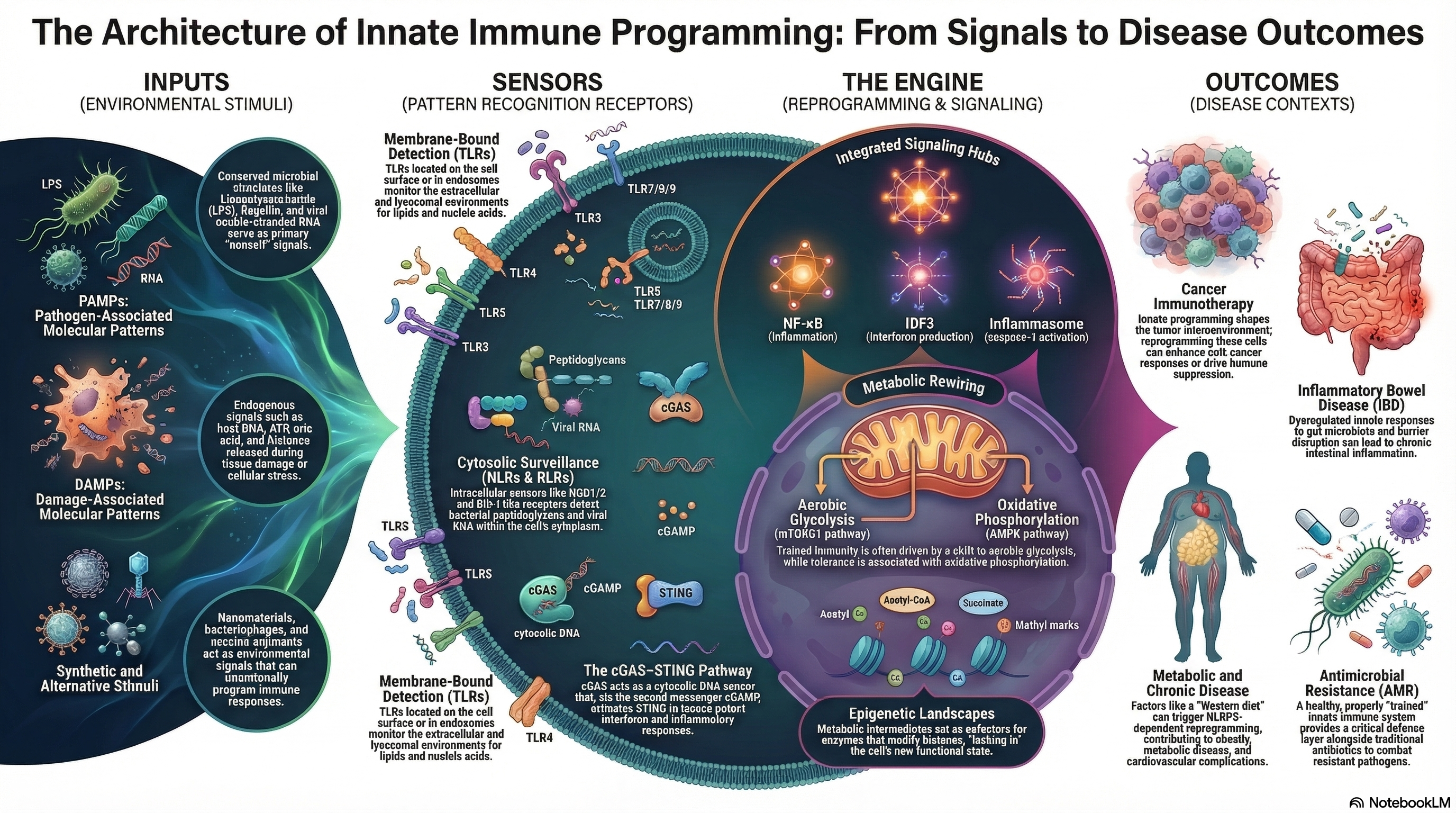
Programmable Innate Immunity
Our core question: Can innate immune responses be deliberately, and safely, programmed into defined protective or functional states?
The innate immune system is traditionally viewed as a rapid but non-specific defence mechanism. However, growing evidence shows that innate immune cells can undergo durable functional reprogramming following exposure to microbial signals, therapeutics, or environmental stimuli. Our research explores whether innate immune responses can be deliberately programmed into defined functional states. These states may enhance host protection against infection, shape responses to therapeutics, or contribute to inflammatory disease.
We investigate the molecular mechanisms that control innate immune programming, including pattern recognition receptor signalling, inflammasome activation, and epigenetic regulation of immune responses. These processes are closely linked to phenomena such as trained immunity, where prior stimulation induces persistent changes in immune cell behaviour through metabolic and chromatin remodelling. Understanding these mechanisms may enable the development of therapeutic strategies that intentionally modulate innate immunity to improve disease prevention and treatment.
Key areas include:
Trained immunity and innate immune memory
Pattern recognition receptor signalling networks
Epigenetic regulation of innate immune responses
Immune programming by microbes, phages, and therapeutics
Engineering sustained innate immune protection
Operating across three interacting layers:
1. Immune sensing - Pattern recognition receptors detect therapeutic particles and microbial signals. These include TLRs, NLRs, and cytosolic DNA sensors that detect viral, bacterial, and nanoparticle stimuli.
2. Cellular reprogramming - Innate immune cells undergo metabolic and epigenetic changes that alter their future responsiveness. These changes include glycolytic rewiring, mitochondrial stress responses, and chromatin modifications associated with trained immunity.
3. Functional outcomes - The resulting immune state determines cytokine production, complement activation, inflammasome signalling, and therapeutic safety. This structure mirrors how trained immunity operates through metabolic and epigenetic reprogramming of innate cells.
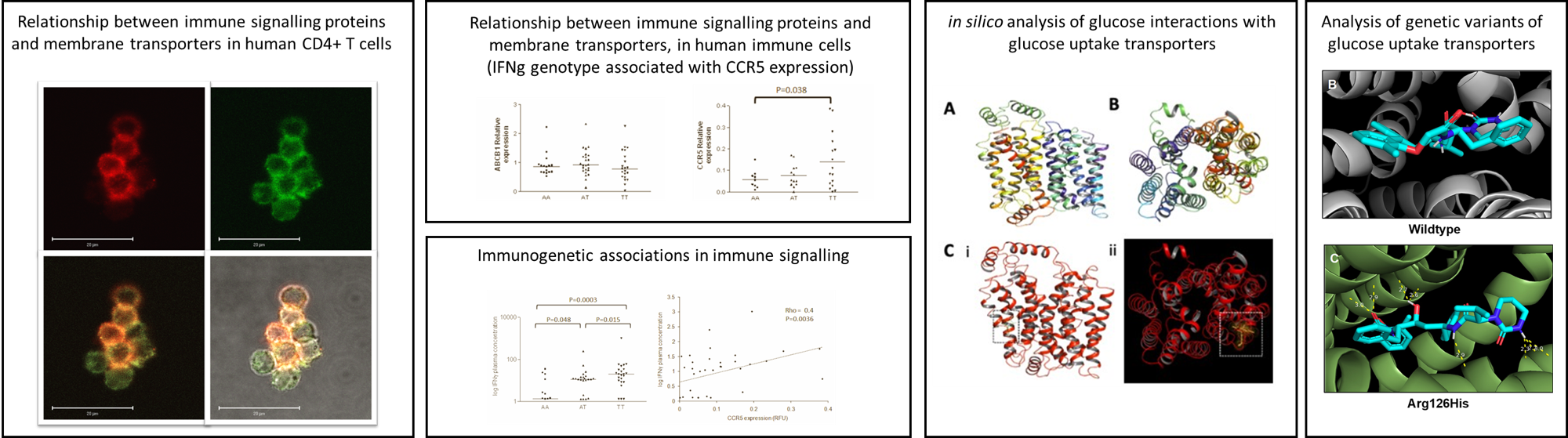
Immunometabolism and Innate Immune Variability
Our core question: How does cellular metabolism or systemic metabolic state shape innate immune responses?
Innate immune responses are strongly influenced by cellular metabolism and bioenergetic state. Changes in glycolysis, mitochondrial activity, and nutrient utilisation can fundamentally alter how immune cells respond to stimulation. Our research investigates how metabolic and bioenergetic pathways shape innate immune function and contribute to interindividual variability in immune responses. These metabolic programmes determine immune cell activation, cytokine production, and inflammatory potential, linking cellular energy metabolism directly to immune regulation.
We study how metabolic state, disease context, and environmental exposures influence innate immune behaviour to understand why individuals respond differently to infection, vaccines, or complex therapeutics.
Key areas include:
Immunometabolism of innate immune cells
Bioenergetic control of inflammatory signalling
Metabolic drivers of trained immunity
Inter-individual variability in immune responses
Metabolic influences on therapeutic safety and efficacy
Keywords: innate immunity, immune metabolism, microenvironment, transporters
Research groups
Research grants
Biologics Regional Innovation and Technology Ecosystem (BRITE)
RESEARCH ENGLAND (UK)
July 2025 - June 2028
Lipid Voyage: Exploring the biology, safety and biotechnological application of lipid-phage complexes found in Breast Milk. (Lipid voyage)
BIOTECHNOLOGY & BIOLOGICAL SCIENCE RESEARCH COUNCIL
September 2024 - August 2027
Rational Engineering of Inorganic Crystals as Novel Therapeutics to Induce Antimicrobial Activity in Host Cells
INFECTION INNOVATION CONSORTIUM (UK)
November 2024 - June 2025
Building a UK IntraCellular Drug Delivery Centre (ICDDC)
INNOVATE UK (UK)
January 2023 - March 2026
A game changer for the treatment of osteoarthritis: a cost effective combined advanced therapy to treat knee osteoarthritis (SINPAIN)
UK RESEARCH AND INNOVATION
May 2022 - October 2026
National Hub for Advanced Long-acting Therapeutics (HALo)
ENGINEERING & PHYSICAL SCIENCES RESEARCH COUNCIL
November 2024 - October 2030
CF- TRAILFINDER (CF- TRAILFINDER)
CYSTIC FIBROSIS TRUST (UK)
December 2024 - November 2029
Automated high-resolution microscope platform with robotics to study pharmacodynamics in 3D cell cultures
MEDICAL RESEARCH COUNCIL
November 2022 - March 2023
Safety Testing In The Life Cycle Of Nanotechnology-Enabled Medical Technologies For Health (SAFE-N-MEDTECH)
EUROPEAN COMMISSION
April 2019 - September 2023
Regulatory Science Framework for Nano(bio)material-based Medical Products and Devices (REFINE)
EUROPEAN COMMISSION
December 2017 - February 2022
Assessing the therapeutic effects of renal regenerative medicine therapies using a suite of in vitro assays
CHILDRENS RESEARCH FUND (UK)
February 2021 - October 2021
European Nanotechnology Characterisation Laboratory
EUROPEAN COMMISSION
May 2015 - December 2019
Developing novel tools and technologies to assess the safety and efficacy of cell-based regenerative medicine therapies, focusing on kidney disease RenalToolBox
EUROPEAN COMMISSION
November 2018 - April 2023
Investigating how cell based regenerative medicine therapies modulate T-cells and macrophages to ameliorate acute kidney injury
KIDNEY RESEARCH UK (UK)
November 2018 - September 2021
Exploring how cell therapies ameliorate renal damage
MERSEY KIDNEY FIRST (UK)
October 2018 - September 2021
Long acting NRTI therapies for HIV
NATIONAL INSTITUTES OF HEALTH (USA)
July 2017 - June 2023
Multi-modal high resolution preclinical PET+SPECT+CT scanner
WELLCOME TRUST (UK)
July 2018 - June 2024
Bioengineered substrates for human pluripotent stem cell culture
WELLCOME TRUST (UK)
November 2016 - October 2018
Investigation of the use of nanotechnology to reduce the side effects associated with antiretroviral protease inhibitors
BRITISH SOCIETY FOR ANTIMICROBIAL CHEMOTHERAPY (UK)
May 2015 - December 2016
Characterising the influence of nanoparticle characteristics on their interference/ interaction with endotoxin
ROYAL SOCIETY (CHARITABLE)
October 2015 - September 2016
Research collaborations
Dr Ishwar Singh
Spatial control of innate immune modulators
Dr Peter Sjo
Innate modulation for viral infection resolution
DNDi
Innate modulation for viral infection resolution
Dr Thom Law
Immune modulating antimicrobials
DSTL
Innate immune modulation to reduce morbidity of bacterial infections.
Prof. Yvonne Perie
Structure-activity relationships between liposomes and complement activation
University of Strathclyde
Dr Rob Vandebriel
Immunotoxicological assessment of nanobiomaterials (REFINE)
RIVM - National Institute for Public Health
Dr Sven Even Borgos
Interindividual variability in Immunotoxicological responses to nanobiomaterials
SINTEF
Dr Joe Vetro
Nucleic acid based nano-therapies for cancer
University of Nebraska Medical Centre
Research partnership to develop, biocompatible, nucleic acid based therapies for drug resistant cancers.
Dr Bryant Nelson
Standardsation of immunotoxicological assessment of nanobiomaterials
National Institutes of Health (NIH and ASTM
Prof. Patricia Murray
Assessment of novel model systems to investigate renal inflammation
Dr Bettina Wilm
Impact of extracellular vesicles on macrophage function and phenotype
Prof, Rachel Williams
Biocompatibility of advanced materials, with nano-topography
Dr Tom McDonald
Development of, immunomodulatory, lipidic nanoparticle delivery systems.
Prof. Raechelle D'Sa
Immunotoxicological assessment of novel nanomaterials for wound healing
Translational pharmacology
translational immunology pharmacology of immune responses immune pharmacology drug immune interactions biological response to therapeutics safety assessment of advanced medicines therapeutic immune modulation drug induced immune responses
Disease and clinical context
infectious disease immunology antiviral immunity respiratory virus immunity pandemic preparedness immunology host defence mechanisms microbiome immune interactions inflammatory disease immunology metabolic inflammation cardiometabolic immune activation neuroinflammation
Prof. Kirill Afonin
Inflammasome recognition of, and response to, nucleic acid-based nanoparticles
UNC Charlotte
Immunocompatibility assessment of, and structural relationships between, nucleic acid nanoparticles.
Biomaterials and drug delivery
biomaterial immune interactions immune responses to biomaterials foreign body response implant immune compatibility drug depot immune responses nanocrystal therapeutics long acting injectable medicines sustained release drug delivery polymer based therapeutics injectable depot systems
Immunocompatibility and immune safety
immunocompatibility immunotoxicology immune safety of therapeutics immune mediated toxicity cytokine release mechanisms complement activation pathways inflammasome signalling immune cell activation profiling innate immune assay development immune responses to complex medicines
Lipid nanoparticle and vaccine research
lipid nanoparticles immunology LNP immunogenicity mRNA vaccine delivery systems RNA vaccine technologies vaccine adjuvant nanomaterials LNP innate immune activation inflammasome activation by nanoparticles immune responses to vaccine carriers vaccine reactogenicity mechanisms
Nanomedicine and advanced therapeutics
nanomedicine nanotherapeutics nanoparticle therapeutics drug delivery nanotechnology lipid nanoparticle delivery mRNA delivery systems RNA therapeutics delivery nucleic acid delivery systems biomaterial based therapeutics synthetic immunomodulators precision biomaterials
Epigenetic and systems regulation
pigenetic regulation of innate immunity histone modification immune regulation chromatin accessibility in immune cells H3K4me3 trained immunity H3K27ac immune activation transcriptional control of innate immunity systems immunology multi omics immunology integrated immunology modelling immune network regulation
Research Themes
Immunocompatibility
programmable innate immunity innate immune programming innate immune reprogramming innate immune memory trained immunity innate immune plasticity innate immune signalling innate immune activation innate immune modulation innate immune regulation host innate immunity innate immune sensing pattern recognition receptor signalling toll like receptor signalling NOD like receptor signalling RIG I signalling cGAS STING signalling innate antiviral immunity innate immune defence host directed immunity
Sustained immune protection concepts
sustained innate immune prophylaxis innate immune prophylaxis long duration innate immunity durable innate immune responses host directed antiviral protection innate immune priming innate immune training strategies long acting immune modulation innate immune preparedness broad spectrum antiviral immunity non specific immune protection
Dr Steve Street
Precision biomaterials
Precision biomaterials for programmable innate immunity
Experimental models and methods
human immune cell models PBMC immune assays THP 1 monocyte models macrophage activation assays multiplex cytokine profiling flow cytometry immunophenotyping Seahorse metabolic analysis metabolomics in immunology immune signalling assays innate immune functional assays
Long acting therapeutics
long acting medicines long acting injectables extended release therapeutics long duration drug delivery depot drug delivery systems sustained release formulations long acting antiviral therapy long acting prophylactic medicines
Immunometabolism and cellular regulation
immunometabolism immune cell metabolism metabolic regulation of immunity glycolysis in immune cells mitochondrial metabolism in immunity TCA cycle metabolites in immunity succinate signalling in macrophages fumarate mediated immune activation acetyl CoA epigenetic regulation metabolic control of inflammation metabolic reprogramming of macrophages bioenergetics of immune cells