Watch the video below to hear from our researchers and industry partners about how we collaborate and the benefits it brings to the Centre and our partners.
The map below shows the breadth of the Centre's collaborations across the world.
UK Collaborations
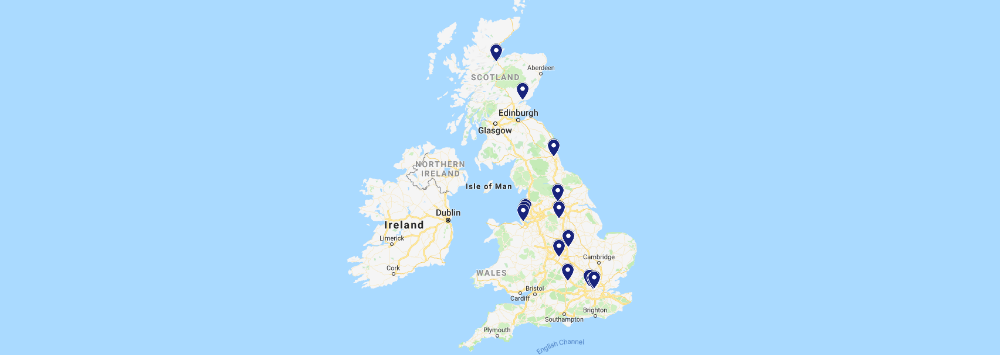
We have many collaborators within the UK, including those from other MRC-funded Centres, Units and Institutes.
MRC Toxicology Unit
We have long-standing ties with the MRC Toxicology Unit, that have typically been built through joint PhD studentships. Through these studentships we have been able to access expertise and technology to support global microRNA analysis of hepatocytes/cholangiocytes (Bushell) and mitochondrial bioenergetics and cell death (McFarlane). More recently, and through the Toxicology Unit’s increased interest in immunopharmacology, we have begun to develop joint areas of interest around immune checkpoint inhibitors.
MRC Centre for Medical Mycology
A number of anti-fungal agents are associated with DILI. We are therefore working with the CMM, and their world-leading expertise in fungal biology and host responses, to investigate mechanisms of DILI toxicity. A joint studentship in this area is expected to start in October 2020.
MRC Centre for Neuropsychiatric Genetics and Genomics
Clozapine is the only licensed medication for treatment-resistant schizophrenia but is associated with a relatively high risk of agranulocytosis, which can be lethal. Our two Centres are working together to combine clinical samples, expertise and resource in order to investigate the genetic and functional factors that drive this serious side effect.
MRC Laboratory of Molecular Biology
We are collaborating with Dr M. Madan Babu’s laboratory which is developing novel computational methods to better understand how regulation is achieved at different scales of complexity. His interest in the pharmacogenomics of drug responses and drug targets in the human population, using multi-scale modelling approaches, are directly relevant to many aspects of CDSS research.
MRC Mitochondrial Biology Unit
There are now many examples of adverse drug reactions in which the mitochondria are targeted. Our research is investigating how individual mitochondrial variation plays a role in the induction of these events, and we are collaborating with Professor Patrick Chinnery and colleagues in the Mitochondrial Biology Unit.
European Collaborations 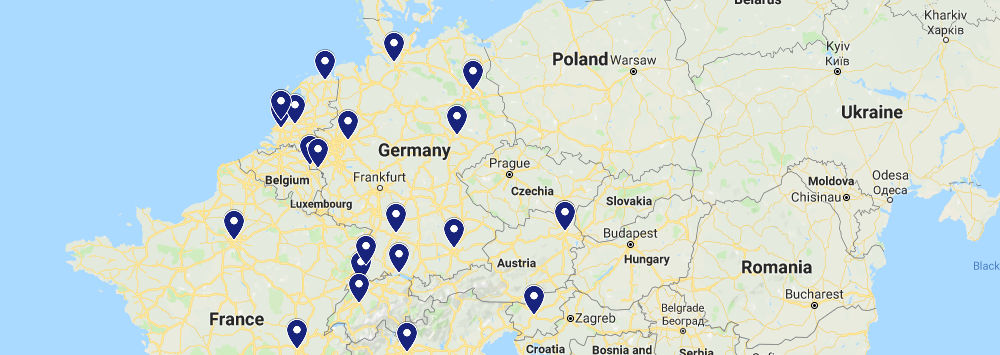
We work with colleagues across Europe, and are particularly active in large H2020 consortia, including those funded through the Innovative Medicines Initiative (IMI).
TransQST
We lead the TransQST project (http://transqst.org/) which is aiming to improve the translation from nonclinical to human safety during clinical trials. With 22 partners (10 academic; 3 SME; 9 pharma) we are integrating PB-PK/PD models with outputs from systems models in order to develop novel Quantitative Systems Toxicology models. The project is working on four distinct target organs for drug-induced injury: liver, kidney, cardiovascular and gastrointestinal-immune systems.
TransBioLine
The Translational Safety Biomarker Pipeline (TransBioLine) Project is a new IMI-funded program to generate exploratory and confirmatory data supporting regulatory qualification and acceptance of novel safety biomarkers for five target organ systems (kidney, liver, pancreas, vascular, and central nervous system) for application in drug development. CDSS scientists are providing drug-induced liver injury and informatics expertise whilst Professor Robert Sutton from UoL is leading the pancreas workpackage.
U-PGx
We are part of the Ubiquitous Pharmacogenomics project (http://upgx.eu/) that is addressing major challenges and obstacles for the implementation of pharmacogenomic (PGx) testing in patient care. Specifically, U-PGx is investigating whether the emerging approach of pre-emptive genotyping of an entire panel of important PGx markers is cost-effective and results in a better outcome for patients.
Rest of the world
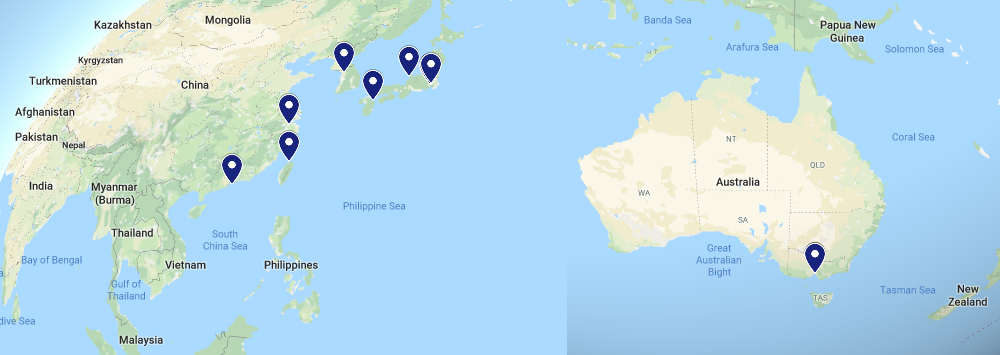
We have many collaborators that are further afield. Some examples include:
Professor Tony Purcell, Monash University, Melbourne, Australia
A two-way collaborative study between the Purcell laboratory and the CDSS has been integral in establishing immunopeptidomics research at the CDSS to explore pathways of drug-specific T-cell activation. Scientists within the Purcell lab hosted students and post-doctoral researchers from the CDSS, to train them in the methods required to perform HLA peptide elution and characterization of the associated peptide repertoire. Upon their return to the CDSS, protocols, cell lines and knowledge on how to quantify the large of amounts data generated were transferred and successfully implemented. As a direct result of this knowledge exchange, the CDSS were awarded an MRC project grant in 2018 to explore the importance of drug metabolism and adduct formation in the activation of drug-specific T-cells.
Click here for more information.
Dr Karen Cohen, University of Cape Town, South Africa
Dr Cohen holds a Newton Advanced Fellowship and is working with us to characterise signature patterns of biomarkers associated with drug-induced liver injury following anti-tuberculosis treatment, which is the second most common cause of adverse drug reaction-related death in South African medical wards. Dr Cohen is also part of our NIHR-funded Global Health Research group on warfarin anticoagulation in patients with cardiovascular disease in Sub-Saharan Africa that aims to improve anticoagulation by developing clinical dosing algorithms, and further improve anticoagulation quality by using novel technological advances.
Click here for more information.
Back to: Centre for Drug Safety Science