Medical device development
The Biomechanical Engineering Group is working on the development of a number of medical devices, most of which aim to improve clinical practice in ophthalmology.
The methodology we adopt is the same in all cases, starting with establishing clinical need, followed by development of concept designs, then assessment of designs using numerical analysis, manufacture of prototypes based on designs that appear to be successful in numerical trials, and finally experimental testing on ex vivo eyes and clinical in vivo validation.
Group studies
A. Contact lens device for continuous monitoring of intraocular pressure
Intraocular pressure (IOP) is the major modifiable risk factor for glaucoma. Current glaucoma management practice relies on measuring IOP during office hours in six-monthly clinic visits, and the measurements are used to indicate the amount of IOP lowering medication to be prescribed until next visit. However, the dynamic nature of IOP, and the fact that it changes with common activities such as physical efforts, drinking and sleeping, makes short term measurements of IOP insufficient.
Efforts have been made for more than 40 years to develop technologies to enable the continuous measurement of IOP. The BioEG has been working since 2008 on the development of a contact lens device that is equipped with an innovative pressure sensor (to measure IOP) and a telemetry system (to transmit the measurements wirelessly to an external instrument). The work has been supported by two grants from the NIHR and is expected to reach clinical validation soon.
B. Contact device for in vivo measurement of corneal biomechanics 
Determination of corneal biomechanical properties in vivo is essential for the accurate planning of refractive surgery, design of contact lenses, optimisation of collagen cross-linking treatment in keratoconus and selection of corneal implants to overcome refractive errors. The current inability to determine corneal biomechanics of the living eye has meant that these applications could not be optimised for individual patient’s needs.
A new device has been developed by the BioEG to measure the in vivo mechanical properties of the human eye. The device has been through numerical analysis, experimental assessment and is to undergo clinical validation soon.
C. Warming device for meibomian gland dysfunction 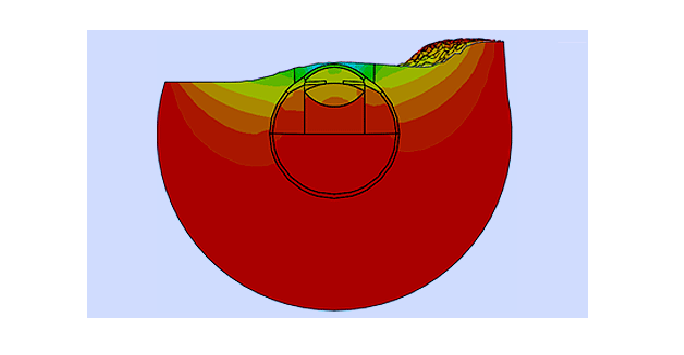
Meibomian Gland Dysfunction (MGD) affects a large percentage of the population and leads to the usual discomforts associated with dry eyes. The condition is usually managed by a combination of heat and massage to melt the natural meibum and allow it to flow onto the surface of the cornea. Current techniques to heat the eye lack precession in the amount and duration of the heat applied.
The BioEG has developed a simple technique to offer tight control over both the magnitude and duration of heating and control over the amount of steam reaching the eye. As the technique was based on representative numerical modelling of the heat flow through the eyelid and the eye, it enables reaching higher temperature than other methods, which is quite important for severe cases of MGD.
D. Non-contact tonometer with more accurate IOP measurements
In eyes with thick and/or stiff tissue, the resistance to deformation is high and as a result the IOP value is over-estimated in tonometry. The resulting error can be serious, leading to either false positives (in cases with normal IOP and thick or stiff tissue) or false negatives (cases with high IOP and thin or compliant tissue). These outcomes can have a detrimental effect on glaucoma management (for false negative cases) or lead to ineffective use of healthcare resources (false positive cases).
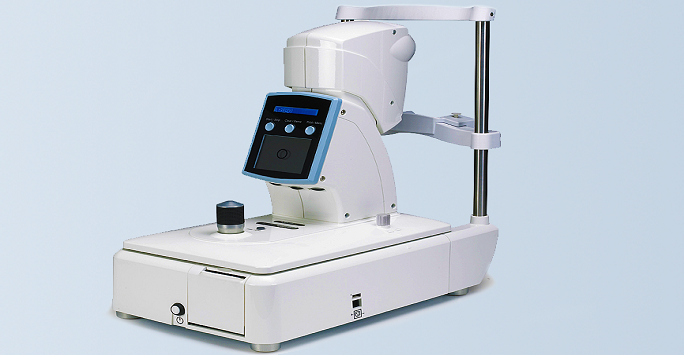
The BioEG has developed a non-contact tonometer in collaboration with Keeler. The tonometer has new technology to monitor corneal deformation under the effect of the air puff. The deformation distribution is then analysed to separate the effects of IOP from that of corneal stiffness. The technology has been embedded in a prototype that has been assessed experimentally on human donor ocular globes and later experimentally at Moorfields Eye Hospital. The results are being prepared for publication. This project was funded by the NIHR.