Sailing towards the island of stability: decay spectroscopy of flerovium
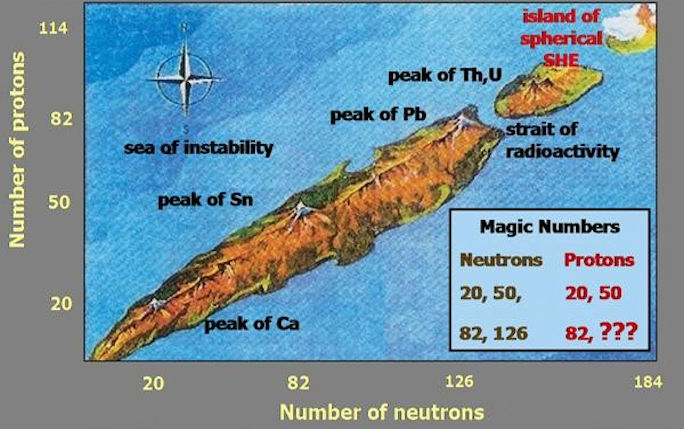
A holy grail of nuclear physics is to understand the stability of the periodic table’s heaviest elements. The heaviest element naturally occurring on earth is uranium, element 92. Its most common isotope uranium-238 consists of 92 protons and 146 neutrons, arranged in shells. Some combinations of protons and neutrons are more stable than others, which gives the nuclei extra stability. These closed shells are characterised by “magic” numbers, i.e. at 2, 8, 20, 50, 82, Examples of such nuclei are oxygen-16, calcium-48 or lead-208. One prediction of the Nuclear Shell Model going back to the 1960s is the emergence of an “Island of Stability” far beyond the elements naturally occurring on earth, centered around the “magic” proton number 114 and neutron number 184, and internationally great efforts are made to map out the location and extent of this island of stability experimentally.
The biggest challenge is posed by the fragility of such heavy nuclei, which leads to production rates of less than a single atom per day. As part of the FAIR Phase-0 programme at the GSI Helmholtzzentrum für Schwerionenphysik, an international collaboration between researchers from Lund, Liverpool, Mainz, Madrid, Jyväskylä, Darmstadt, Cologne, Berkeley, Livermore and GSI have bombarded a plutonium-244 target with 6x1018 nuclei of calcium-48. The target had to be rotated to avoid it melting in the very intense ion beam. We were able to produce ~30 atoms of flerovium (Z=114) and study their decays.
The nuclei produced then undergo a series of fast alpha decays while implanted in a dedicated detector system based on the original Liverpool/Lund TASISpec spectrometer. A careful analysis of the decays can reveal the evolution of shape and structure along these decay chains.
Like many voyages of discovery the primary destination also leads to some unexpected intermediate stops, and this experiment is no exception. The observed decays from flerovium-288 (Z=114) first into copernicium-284 (Z=112) led to the first observation of a decay into the newly discovered isotope darmstadtium-280 (Z=110).
The really surprising finding of this study was the observation of an excited state in one of the produced copernicium-282 isotopes. We did not expect to be able to see such a state as alpha decays between even-even elements are usually populating the ground state. Together with theoretical calculations the experiments showed that the observed decay properties were not compatible with the hypothesis that the island of stability is centered on flerovium.
The article is available (open access) here: https://journals.aps.org/prl/abstract/10.1103/PhysRevLett.126.032503