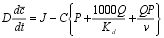
|
Pollutant records in deep lakes |
Return |
| A great water depth results in a significant water column reservoir of dissolved ions. This has the effect of weakening the coupling between influx of a component, and its transfer rate to the sediment. A mass balance model can be used to constrain this effect. The model outlined below is discussed further elsewhere: | |
| Inorganic geochemical methods in palaeolimnology. J.F. Boyle. In (Eds) W.M. Last & J. Smol, Tracking environmental change using lake sediments: physical and chemical techniques (2001). Kluwer Academic, Dordrecht. ISBN 0-7923-6482-1 | |
| The metal mass balance for unit area of lake bed depends on the competing input and output fluxes. The input flux, J, is balanced by burial, outflow in dissolved form, and outflow as suspended solids. Each of these outputs can be expressed in terms of the metal concentration on particles, C. The mass balance can thus be expressed: |
| 1 | 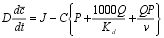 |
where | D = | depth (m) |
| c = | total water held metal, dissolved and particulate (mg m-3) | |||
| J = | areal metal input (mg m-2 yr-1) | |||
| C = | metal bound to particles (mg kg-1 ) | |||
| P = | areal particle flux (kg m-2 yr-1) | |||
| Q = | areal water flux (m3 m-2 yr-1) | |||
| Kd = | distribution coefficient (m3 tonne-1 ) | |||
| v = | settling velocity (m yr-1 ) |
| To solve this expression, the fluxes in the lake must be expressed in terms of the mean water held metal, c , rather than concentration on particles. c is given by: | 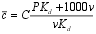 |
2 |
| To make the solution more straightforward, the mass balance can be expressed in terms of c | 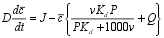 |
3 |
| A large part of this expression will be constant for a given lake, under constant sediment flux, and can be replaced by a single parameter, k, where k is given by | 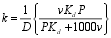 |
4 |
| This gives the final equation for variation in c., where ce is given by expression 6. | 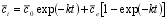 |
5 |
| Expressions 5. and 6. can be solved using a finite difference approach, for changing values if the metal influx, J | 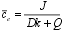 |
6 |
|
Application of the model | |
| This model can be used to predict the effect of water depth on the link between trace element flux and sediment composition. The diagrams below consider hypothetical impacts of changes in the lake system on the sediment concentration of a trace element, X. |
|
Impact of changes in the supply rate of X to the lake. | |
| This is an important case, as it is common to use lake sediment records to infer changes in the supply fluxes of trace elements. The result is critically dependent on how strong the trace element binds to the sedimenting particles (measured here by Kd). For high Kd values (Log Kd = 6.5) typical of Pb, we see that a five year period of enhanced X flux leaves an almost perfect record in a shallow lake (10 m), and a clear, if somewhat distorted record in a deep lake. | 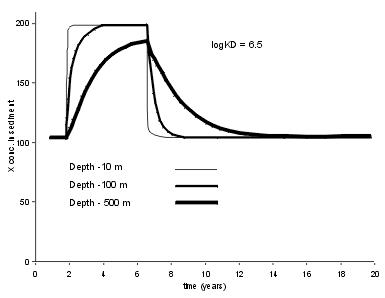 |
| For a less strongly bound trace element the results are different. At Log Kd = 5.0, typical for Zn, we see that a five year period of enhanced X flux leaves a good record in a shallow lake (10 m), but an indistinguishable record in the 500 m deep lake. | 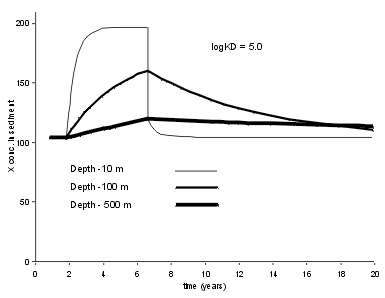 |
|
Impact of changes in the particle accumulation rate in the lake sediment. | |
| The insensitivity of the sediment record of deep lakes to changes in trace element supply, does not mean that the trace element concentration profile in the sediment cannot show sharp changes. A change in the particle deposition rate will cause an instantaneous change in sediment composition through dilution. To the right we see the effect of doubling the particle flux in the lake; this pretty much halves the sediment concentration of X for each of the water depths, as long the Kd is high. | 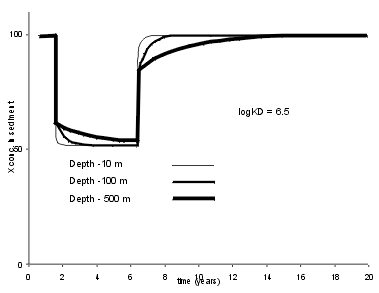 |
| For low Kd values, this dilution effect is reduced by the high water column trace element inventory. There is still a good signal in the shallow lake, but the deep lake produces a very smoothed record. This means that in deep lakes it is important not to infer external trace element supply simply using sediment trace element accumulation rates. |  |
|
Impact of changes in Kd. | |
|
Abrupt changes in sediment composition can also be caused by changes in Kd. An increase in Kd can result from an increased proportion of algal matter, while a decrease in Kd can be caused by increasing calcium carbonate.
Increasing the Kd causes a near instantaneous increase in trace element concentration because the particles can take elements from the water column reservoir. Of course, this reservoir is not inexhaustible, and eventually a new steady state will be reached in which the concentrations return approximately to their pre-change values (not quite the same, as the change in Kd will have altered the outflow losses). This effect increases with the solubility of the trace element, and the depth of the lake. In the 10 m deep lake, for the Kd values typical for Pb (c. 106.4), we see that the increase in concentration is very short lived; the particles take more Pb, but rapidly deplete the reservoir and establish a new equilibrium. When the Kd returns to normal the reverse happens. In a shallow lake, and with typical sedimentation rates, sediment mixing, and sampling intervals, such an event is probably undetectable for Pb. | 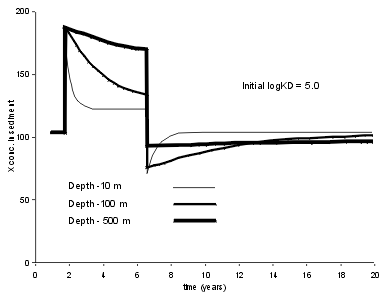 |