New insights into protein nanomachine assembly in pathogenic bacteria
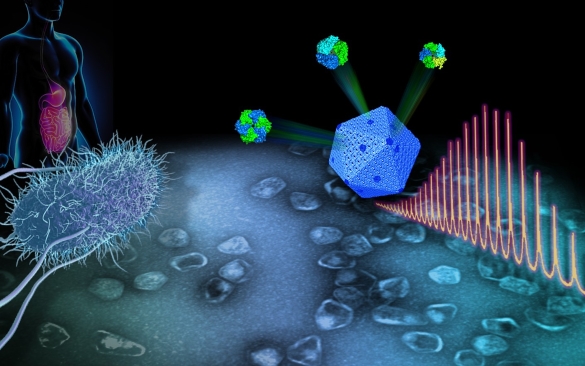
Scientists at the University of Liverpool have provided a clear view of the composition and structure of a self-assembling protein organelle, which is essential for the growth of pathogenic bacteria.
A new study, published in Nature Communications, reveals how hundreds of proteins self-assemble to build an active nanoreactor in Salmonella to boost metabolism.
Many pathogenic bacteria, such as Salmonella, use protein-formed nanomachines, called bacterial microcompartments, to metabolise diverse nutritional sources and help them thrive in human gut tissues. The bacterial microcompartment is a specialised organelle composed of a nanoscale protein shell filled with multiple metabolic enzymes. The shell structure is made up of a series of protein ‘tiles’ that slot together precisely.
“The bacterial microcompartment is an amazing naturally-occurring system that is purely made by proteins possessing different structure and functions. These proteins recognise and bind with each other in cells to form a highly ordered nanostructure,” said Professor Luning Liu from the University’s Institute of Integrative Biology, who led the research. “We are just starting to understand the real organisation of the bacterial machines.”
In collaboration with Professor Rob Beynon at the Centre for Proteome Research and Professor Jay Hinton at the Institute of Integrative Biology, the researchers used state-of-the-art proteomic techniques to study the exact abundance of individual building proteins and how they assemble and organise to build a functional organelle involved in the propanediol degradation in Salmonella.
Their findings also reveal that the protein composition and overall structure of the propanediol-utilising microcompartment are adjustable, so their metabolic performance can be regulated in cells.
“Our results help us better understand how bacterial organelles are formed and regulated to execute biological catalysis,” added Professor Liu. “By using the natural building principles, researchers can make proper designs to generate artificial and custom metabolic machines or nanoreactors, with the ultimate goal of boosting production of valuable compounds.”
The project was funded by Royal Society, Biotechnology and Biological Sciences Research Council (BBSRC), Wellcome Trust, and the China Scholarship Council. The project was also carried out in collaboration with the Centre for Cell Imaging at the Institute of Integrative Biology and Liverpool Electron Microscopy.
Research reference
The paper ‘Decoding the stoichiometric composition and organisation of bacterial metabolosomes’ is published in Nature Communications 2020, DOI: 10.1038/s41467-020-15888-4.